
See why we’re trusted by healthcare organizations across the country.
Learn more
The healthcare industry experienced an extraordinary amount of regulatory activity last year, with the COVID-19 pandemic accelerating some efforts and delaying others. Here’s a look at the key regulatory developments impacting the Surescripts Network Alliance in 2021.
Under the current Centers for Medicare & Medicaid Services (CMS) regulations, beginning January 1, 2021, Part D plans were required to implement at least one real-time benefit tool capable of integrating with providers’ e-prescribing and electronic health record (EHR) systems.
Despite the disruption to the congressional schedule due to the pandemic, the law was passed by Congress late last year. It includes a “trifecta” of efforts to accelerate the adoption and use of real-time benefit tools (RTBTs) by:
The Office of the National Coordinator for Health Information Technology's (ONC) certification and physician payment incentive will be effective by January 1, 2022. The Part D insurer provision is already in effect.
Surescripts Real-Time Prescription Benefit meets the requirements of this CMS mandate, and helps providers and patients more closely collaborate on choosing a medication that is both effective and affordable. The technology displays less expensive alternatives based on the patient’s benefit plan, so that they aren’t surprised by the cost of their medication at the pharmacy. This can help prevent medication non-adherence and the resulting negative impacts on patient health outcomes.
As of the end of 2020, more than 400,000 prescribers were already using Surescripts Real-Time Prescription Benefit to lower patients’ out-of-pocket costs and prevent “sticker shock” at the pharmacy, which can cause patients to abandon their prescriptions and potentially experience worsening health conditions.
As Dr. Michelle Forbes, a partner and practicing pediatrician at MD Pediatric Associates in north Texas shared, “The technology brings the decision-making back into the exam room, which is a very sacred space. Real-Time Prescription Benefit allows us to decide together, myself and my patient and their parents, the best course of action. Technology brings the decision back where it belongs."
Also at the end of 2020, CMS issued its final rule requiring use of NCPDP SCRIPT Standard v2017071 for all Part D electronic prior authorizations transactions effective January 1, 2021, with enforcement mechanisms in place by January 1, 2022. These rules spur broader adoption and utilization of e-prescribing, which contributes to increased medication adherence and improved patient safety. And electronic prior authorization can speed time to therapy by preventing the “administrative swirl” that so often hinders the manual phone and fax-based process.
Also in December, CMS announced in a final rule its decision to require that Part D providers use electronic prescribing for controlled substances (EPCS) effective January 1, 2021, pursuant to the requirements of the SUPPORT Act of 2018. Despite considering a delay to the mandate until next year, CMS reversed course after reviewing comments from Surescripts and others. We will be watching for CMS to publish a second rule regarding penalties and compliance sometime this year with an effective date of January 1, 2022.
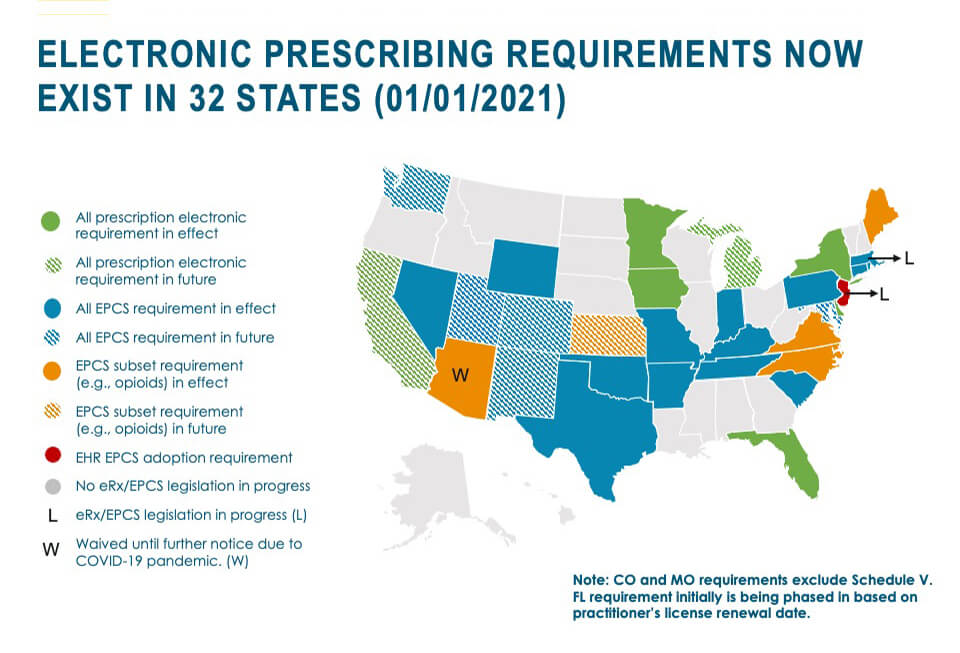
The good news? These mandates work. States with e-prescribing/EPCS mandates saw the largest increases in prescriber adoption in 2019, and we expect legislative actions to continue to serve as catalysts for positive change. Using EPCS, providers can securely prescribe opioid pain medications and other categories of controlled substances to patients in need while preventing fraud and abuse. And these benefits are more timely and crucial than ever given the COVID-19 pandemic and its worsening effect on mental health and the nation’s opioid epidemic.
Last year, the U.S. Department of Health and Human Services (HHS) published the 21st Century Cures Act Final Rule related to improving patients' access to their health information through the exchange of electronic health information. Due to the COVID-19 pandemic, HHS extended the compliance deadline to April 5, 2021.
The rule on Interoperability, Information Blocking and the ONC Health IT Certification Program, issued by the ONC, is intended to clarify and help realize the goals of the 21st Century Cures Act, which was enacted with bipartisan support in 2016.
Surescripts supports the goal of the rule, as we continue to enable the secure exchange of health information to improve patient safety, lower costs and ensure quality care. We are active participants in the work happening across the industry and government to continue to make healthcare information accessible to support patient care.
Never has our network, and the incredible collaboration that makes it possible, been more critical to our nation’s ability to combat and recover from the COVID-19 pandemic. And we remain committed to serving the Surescripts Network Alliance and the millions of healthcare professionals who rely on our network and the technologies it enables every day.
Subscribe to Intelligence in Action to stay informed and engaged with the issues impacting healthcare.